

The use of CO2 as an additive for ignition delay and pollutant control in CH4/air autoignition
E.A. Tingas, H.G. Im, D.C. Kyritsis, D.A. Goussis
Fuel 211, pp. 898-905, (2018)
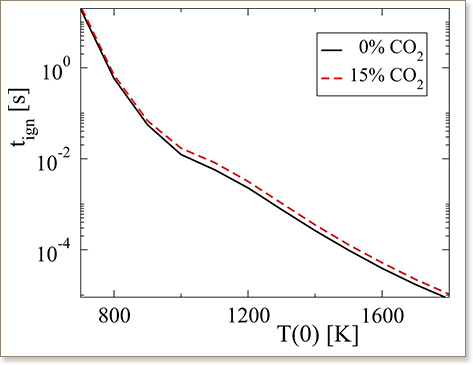
The effect of CO2 dilution on the adiabatic and isochoric autoignition of CH4/air mixtures is analyzed with Computational Singular Perturbation(CSP) algorithmic tools, with a particular emphasis on the determination of the features of the chemical dynamics that control ignition delay and emission formation. Increasing CO2 dilution causes longer ignition delays, lower final temperatures and decreased formation of NO and CO. These effects of CO2 dilution are shown to be entirely thermal, contrary to what happens with dilution with H2O, which also has chemical activity and can reduce ignition delay. For the same initial mole fraction of the diluent, the decrease in final temperature and in NO concentration is larger in the CO2 case whereas the decrease in CO is larger in the H2O case. The thermal effect of CO2 is entirely analogous with those of dilution with the chemically inert Ar, only stronger for the same percentage of initial dilution, because of the larger specific heat of CO2. The reactions that have the largest contribution to the characteristic explosive time scale of the system during ignition delay (H2O2(+M) → OH + OH(+M), CH3O2 + CH2O → CH3O2H + HCO, CH4 + CH3O2 → CH3 + CH3O2H, H + O2 → O + OH, etc.) are not substantially affected by CO2 dilution, neither are the species that are pointed by CSP (CH3O2, H2O2, CH2O, etc.) as having the largest impact on the this timescale. The same holds for the modes that control CO and NO formation. The results point to the possibility of cold exhaust gas recirculation being used in order to produce mixtures with longer ignition delays and therefore substantial resistance to uncontrolled ignition.